Density Golchin
Description and working steps
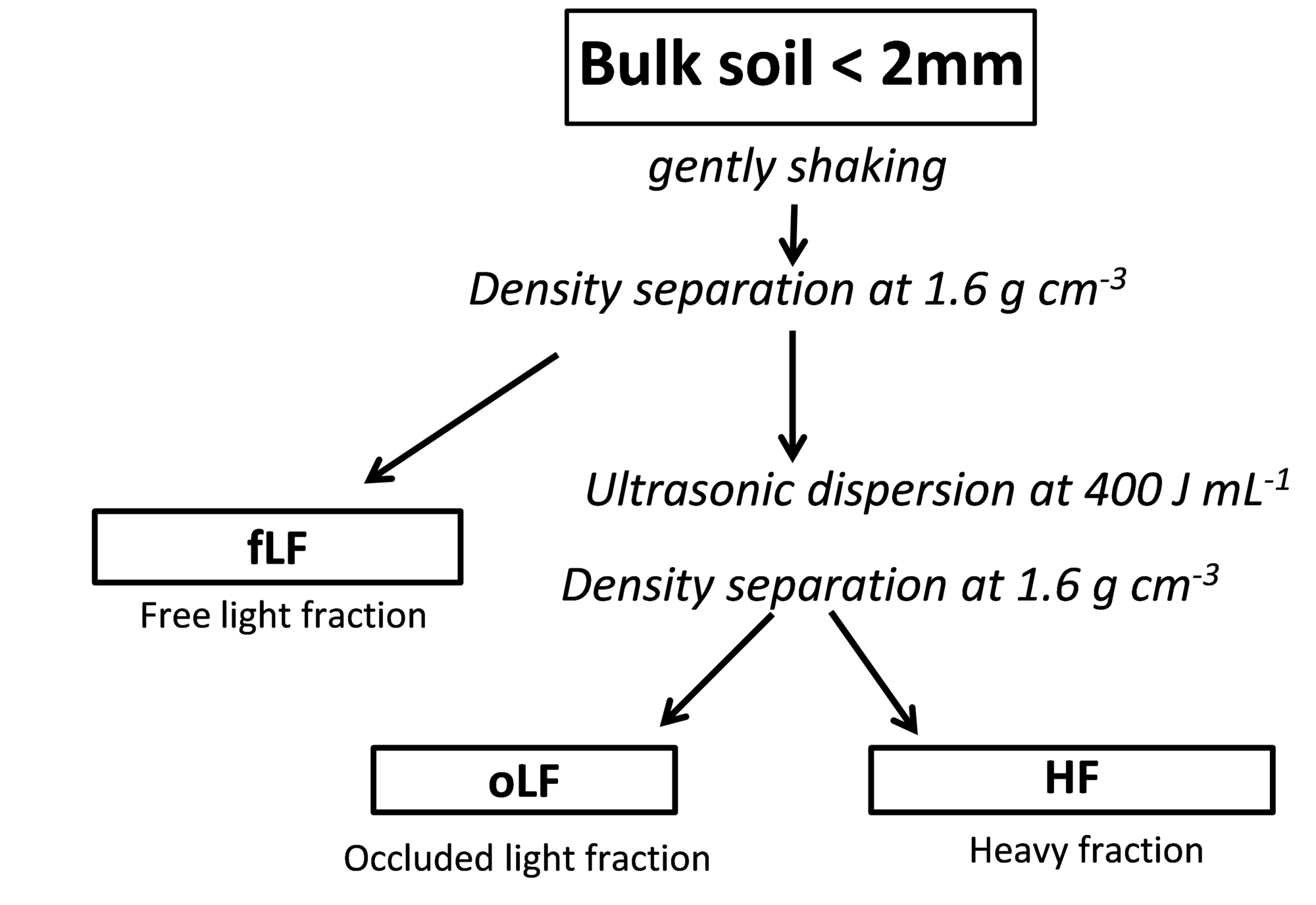
This method is based on the work of Golchin et al. (1994). The aim of this method is to separate the ‘light’ from the ‘mineral-associated’ organic fractions and dividing the light fraction into two different types of light fractions (particulate organic matter): i) the free light fraction (fLF) that mainly consist of large undecomposed or partly decomposed roots and plant fragments and ii) the occluded light fraction (oLF) which consists of much smaller identifiable components such as small partly incompletely composed organic particles such as pollen grain. The oLF fraction is considered an old carbon pool that have been accreted in aggregates during decades of root growth and is that fraction that is must easily lost with soil cultivation.
1. Step: Sample preparation and first dispersion step
The sample need to be sieved through 2 mm and dried. A 250 mL centrifugation tube (glass) is filled with 200 mL SPT solution with a density of 1.6 g mL-1 using a disperser. A subsample of 20 g dry matter fine soil is given into the solution and the tube is closed with a plastic stopper. The tube is gently inverted five times by hand. The particles which adhered to the stopper and the tube were washed into the suspension using SPT solution.
2. Step: Centrifugation
The suspension is allowed to stand for 30 Minutes before centrifugation. Centrifugation is performed at 2000g for 30 minutes to produce a firm pellet. Clay-rich samples may require centrifugation times up to 60 minutes.
3. Step: Separation of the free light fraction
The supernatant with floating particles is poured into a Millipore filter funnel fitted with a washed membrane filter (0.45) and filtered under vacuum. Sodium polytungstate was removed from the organic matter by washing with at least 500 mL deionized water until rinsed water reached and electric conductivity <10 µS/cm. The separated fraction rinsed from the filter using deionised water in a syringe into a small evaporation dish. The rinse water was evaporated by 60°C to obtain the free light fraction.
4. Step: Separation of the occluded light fraction
After removal of the free light fraction the residual soil was suspended again in 200 mL SPT. In order to suspend the soil and destroy aggregates sonification is applied with 450 J/mL. The time for sonification depends on the sonification system and need to be calibrated (link dispersion). Always the highest power output shall be used (Poeplau and Don, 2014). The probe tip is positioned in the middle of the beaker and 1.5 cm below the surface of the suspension. An ice-bath is used to keep the suspension temperature below 60°C. The suspension was allowed to stand for 30 minutes before centrifugation (2000g, 30 minutes). The occluded light fraction was filtered, washed and dried as described above for the free light fraction. The separation of free and occluded light fraction was replicated at least four times for each sample and the replicates of each fraction were combined for analysis.
5. Step: Washing of the heavy fraction
The pellet with the heavy fraction was washed with 250 to 300 mL deionised water after removing the supernatant SPT. In order to get the pellet into a water suspension a Vortex maybe used. The suspension is centrifuged again (2000g, 30 minutes). The washing procedure is repeated at least two times to remove SPT from the heavy fraction. Finally, the heavy fraction is transferred to an evaporation dish and water is evaporated at 60°C.
Initial aim
The aims was to develop a simple method to separate organo-mineral associations (heavy fraction) from partly decomposed products (light fraction) and to separate light fractions into to fractions that derive from different position within the soil matrix (from within aggregates and outside).
Advantages
This method distinguish the two main types of SOC: the mineral bound SOC in the heavy fraction and the particulate organic carbon in two light fractions. The method is widely used allowing comparisons beyond the own study (Don et al., 2009; Schrumpf et al., 2013b).
Disadvantages
The procedure is rather time consuming, it requires chemicals (SPT) and differences between oLF and fLF in carbon turnover times are rather small (Poeplau et al, under review). The separation of oLF from fLF makes sense only in soil that contains aggregates.
References
Golchin A , Oades JM , Skjemstad JO Clarke P (1994) Study of free and occluded particulate organic matter in soils by solid state 13C Cp/MAS NMR spectroscopy and scanning electron microscopy. Soil Research 32, 285-309. (method description)
Don, A., Scholten, T. & Schulze, E.-D. (2009) Conversion of cropland into grassland: Implications for soil organic-carbon stocks in two soils with different texture. Journal of Plant Nutrition and Soil Science-Zeitschrift Fur Pflanzenernahrung Und Bodenkunde, 172, 53-62.
Poeplau, C. & Don, A. (2014) Effect of ultrasonic power on soil organic carbon fractions. Journal of Plant Nutrition and Soil Science, 177, 137-140.
Schrumpf, M., Kaiser, K., Guggenberger, G., Persson, T., Kogel-Knabner, I. & Schulze, E.D. (2013) Storage and stability of organic carbon in soils as related to depth, occlusion within aggregates, and attachment to minerals. Biogeosciences, 10, 1675-1691.